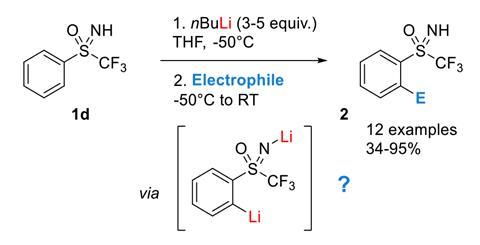
Why do ortho lithiation reactions require a huge excess of butyllithium? | News | Chemistry World | Chemistry World

Directed ortho‐Metalation of Aryl Amides, O‐Carbamates, and Methoxymethoxy Systems: Directed Metalation Group Competition and Cooperation - Miah - 2018 - European Journal of Organic Chemistry - Wiley Online Library

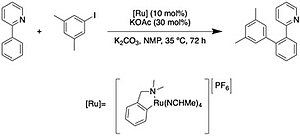
Directed Remote Aromatic Metalations: Mechanisms and Driving Forces - Tilly - 2012 - Chemistry – A European Journal - Wiley Online Library

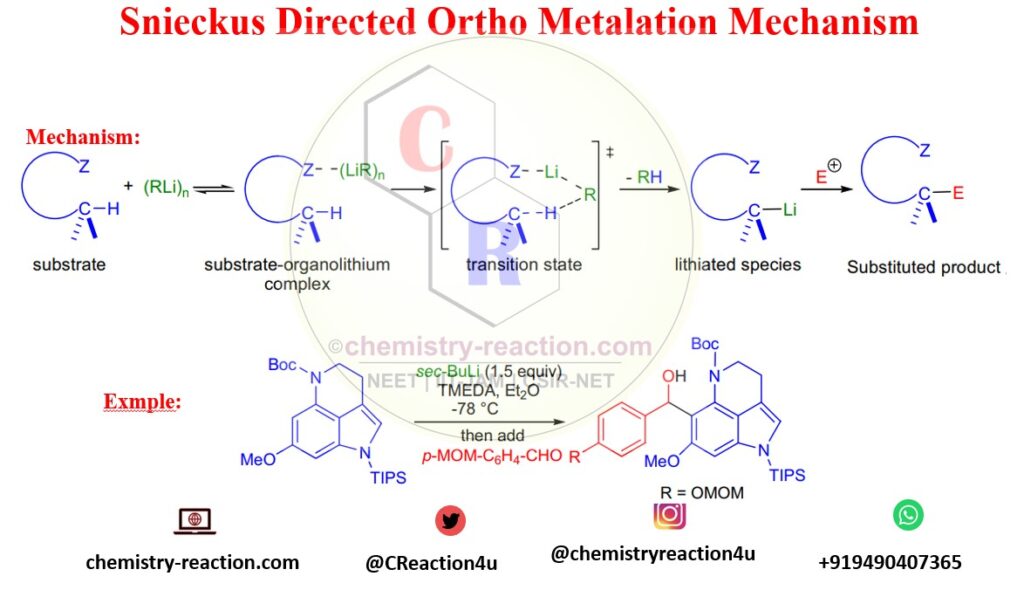
Directed Ortho Metalation in 2021: A Tribute to Victor Snieckus (August 1, 1937–December 18, 2020) | Organic Process Research & Development

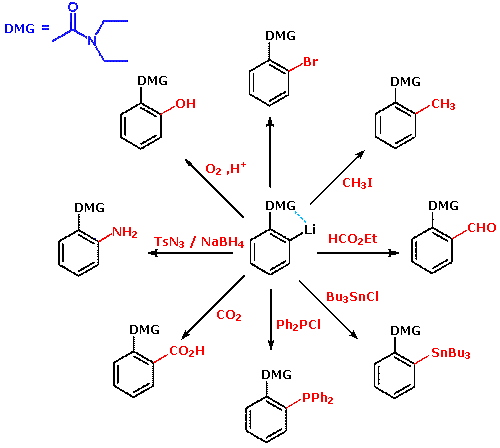
Directed ortho‐Metalation of O‐Aryl N,N‐Dialkylcarbamates: Methodology, Anionic ortho‐Fries Rearrangement, and Lateral Metalation - Miah - 2018 - European Journal of Organic Chemistry - Wiley Online Library

IJMS | Free Full-Text | Interplay between Carbonic Anhydrases and Metallothioneins: Structural Control of Metalation

On the Mechanism of the Directed ortho and Remote Metalation Reactions of N,N-Dialkylbiphenyl 2-carboxamides | Organic Letters

On the Mechanism of the Directed ortho and Remote Metalation Reactions of N,N-Dialkylbiphenyl 2-carboxamides | Organic Letters